Introduction
Within the medical technology and health care devices arena, design and project management professionals must confront a crucial problem. It is that even a relatively small precision metal or plastic part used within a medical device or health care tool — a surgical instrument handle, drug dispenser pump, or diagnostics equipment connection — can cause the failure of the entire unit owing to micrometer-level variations in manufacturing or material defects.
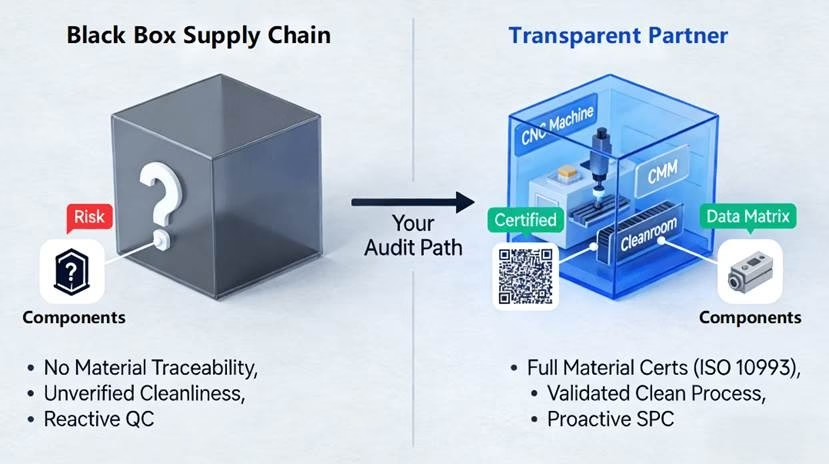
In many cases, the origin of the problem is found within a supply chain “black box”. Procurement strategies based primarily on cost per unit may fail to consider visibility of the manufacturing process, efficiency of the quality control system, and system understanding of the medical grade criteria on the side of the supplier. Such a mismatch can make quality risks hard to anticipate and mitigate during the product design stage. The objective of this article is to introduce the concept of a “quality-first” approach for supplier assessment and partnership development. It will become clear in what way in the medical and healthcare industries choosing an outsourcing CNC precision turning service provider means extending your quality assurance department into a risk-taking one.
H2: What Can Be Done to Avoid Expensive and Potentially Hazardous Manufacturing Issues Through Early Collaborative “Design for Healthcare”?
Design of a medical device goes beyond functional considerations. It needs to be fine-tuned for proper sterilization, biocompatibility, long-term performance, and reproducible manufacturing. Failure to consider tooling access may yield a surface finish with potential contaminants. Variable wall thickness may cause stress concentration resulting in fatigue failure. Early collaboration with a highly experienced medical device manufacturer in design for manufacturability (DFM) analysis is crucial for identifying such risks during the drawing phase. This practice of incorporating manufacturing knowledge upfront represents a fundamental aspect of preventative quality philosophy in the healthcare product development cycle. For those interested in fully understanding the entire process involved in reducing manufacturing risks from design inception, a comprehensive guide to selecting CNC turning parts suppliers offers a holistic perspective, ranging from design guidelines to evaluation checklists..
l Design for Manufacturing and Cleanability: Although something that looks great on a computer-assisted design model could turn out to be difficult to manufacture or impossible to clean because of its complex structure (internal sharp corners, for example). The manufacturing company, having extensive experience in their area, can provide suggestions concerning the necessary adjustments to make it simpler and cleaner, including sufficient draft, large radius of fillets, smooth internal transitions, and so on. This will not compromise the functionality but increase the device’s actual reliability and efficiency, one of the basics of health solutions customization.
l Optimal Dimensions for Performance and Safety: It is essential that the medical device works properly for the whole period of time and does not get damaged by sterilization and possible other stresses. Therefore, the design should ensure the strength of parts and materials under any circumstances and should be made from materials that are compatible with each other. Application experience allows for making appropriate suggestions regarding proper wall thickness and alternative material grade in order to avoid any problems in the future.
l Achieving Process Capability and Design Validation: If you have a tight tolerance on your drawing, but you cannot achieve it with your manufacturing process, then it simply doesn’t matter. A true manufacturing partner will analyze your process capability in the early stages of design and will ensure that you have chosen tolerances that have enough process capability (i.e., a Cpk greater than 1.33). They can even help you determine which of your tolerances need to be kept tighter and which ones may be loosened to make your product more easily manufactured and less expensive.
Why Is Material Traceability More Important Than Cost for Medical Grade Components?
Materials used for medical equipment differ from other engineering products in the sense that they need to not only meet the legal requirements but also be safe for the patients. Besides the mechanical properties of the material, traceability of the material source should be a priority in your choice. A reliable supplier will provide material test certificates from the exact melt or batch. For those implants intended to be inside the human body for a while, you will need ISO 10993 biocompatibility test results. Two pieces of 316L stainless steel made at the same time can have different levels of inclusions or traces, which may influence their corrosion resistance and biocompatibility.
1. The Need for Total Material Traceability
If there’s ever an audit or a problem in the field, you need to be able to trace the part back to the source. You need a closed-loop system of materials management with the supplier that can prove the material grade, material makeup, material properties, and certifications by the supplier. Material traceability is critical for you to demonstrate due diligence. It’s mandatory under quality protocols and your best way of protecting yourself. If a supplier can’t do that, it makes little difference how good a deal their price is.
2. Validating Biocompatibility and Sterilization Compatibility
The definition of “medical grade” titanium or PEEK is not enough. It should be tested according to the purpose (surface device, communicating, or implanting), as well as its contact time with the organism. The manufacturer needs to know about such types of equipment and be able to obtain the corresponding ISO 10993 certifications. They should also provide machining process of material and its cleaning process not to change material properties or to make any residue that will ruin biocompatibility and effectiveness of EtO and gamma sterilization.
3. Providing Material Consistency with Proper Material Management
The consistency of materials is very important. An experienced partner provides only materials with the appropriate certification issued by certified mills that apply strict quality control measures. They also have procedures on the proper handling and storing of materials to avoid contamination, mixing up, or degradation. This approach to the material science and management is one of the key factors for sustainable health practice and the basis of our precision CNC turning.
Is Your Supplier’s “Cleanroom” Really Suited for Your Particular Component?
“Clean manufacturing” of medical components is an evolutionary scale rather than a definitive status quo. Not every part requires a Class 7 cleanroom as its production requirement, but all parts demand controlled environments from their manufacturing process. What really matters here is matching the environmental controls of the supplier to the particular function of the part and the resulting risks involved. The non-critical exterior housing, for instance, is different from an internal joint implant or another component used for a fluid path.
1. Identifying the Right Level of Environmental Control
It starts with the identification of risk factors. What are the possible contamination sources for the device? In the case of implantable devices and surgical instruments, stringent control on particulates and microbial levels is required, which might call for machining in clean rooms. If the device is used for in vitro diagnostic applications, the most important aspect would be biological cross contamination. The right supplier will help you identify your cleanliness requirement, instead of providing blanket cleanroom service.
2. Validation and Monitoring of the Controlled Environment
Environmental hygiene can be attested by the supplier not only through the presence of such features but also by the supplier’s readiness to validate and continuously check their environment. Request the supplier to produce their environmental monitoring plan and also ask what actions are carried out to measure particle count temperature humidity, and pressure differences. Besides, ask about how personnel compliance is monitored and how training is carried out in wearing proper attire for working in the cleanroom; also, request documentation of cleaning and disinfection activities. Environmental monitoring historical reports may be used as evidence of controlled and stable processes.
3. Cleanliness Throughout the Whole Process Flow
Cleanliness has to be integrated in all stages of manufacturing. Thus, the supplier will have to prove that they use biocompatible coolant and cleaning agent in machining. Furthermore, suppliers need to prove that their cleaning processes are validated and efficacious, and they use appropriate cleanroom packaging to maintain part cleanliness after machining. It is important to note that a well-structured environment control procedure can be an indicator of the supplier’s innovation in health technology.
What Can You Look for to Auditing the Supplier’s Quality System Besides ISO 13485 Certificate?
ISO 13485 certificate serves as a minimum threshold to demonstrate that a company has its quality management system implemented. However, it does not mean that the certificate can replace actual checking whether the system operates. When auditing a quality management system, it is essential to look beyond the certificate. It implies studying the way the system works, namely how effectively processes operate, whether there is evidence of continuous improvements, etc. The goal is to investigate whether the suppliers actually implement the system rather than use its documents.
1. Investigation of Internal Audit and Corrective Action Implementation
The supplier’s internal audit results along with the corresponding corrective and preventive action documentation serve as the key elements of your interest. First, you need to determine how effective audits are performed. Are the problems revealed in audits significant or are they superficial? Do the measures taken by the company lead to real improvements or are they temporary solutions? The presence of such a closed-loop system that will identify problems, solve them, and prove that the problem has been addressed proves high maturity level.
2. Evaluating Calibration, Inspection, and Documentation
Once you have seen the plant, check if the measurement instruments like micrometers CMMs gauges, etc. are calibrated. Are these instruments calibrated and still within their calibration validity period? Watch the way of recording inspection results. Is it an integral and on-the-spot recording or just some scribbling on paper? Look into a few DHRs or FAIRs. Are they completed and understandable? Do they provide traceability? It is the actual working in the factory of the documented procedures which decides the quality system’s capacit.
3. Evaluating Change Management and Training
The key to medical device manufacturing is controlled change. Ask to review their process for engineering change orders. What are their process for evaluating, validating, and documenting changes? Additionally, take a look at their employee training. Is it comprehensive and includes the required knowledge related to medical device manufacturing, controlled document handling, and other relevant topics? The supplier that doesn’t treat their changes seriously or has no training program can be risky for your business continuity. A serious CNC turning manufacturer for the medical industry will handle everything professionally.
From Surgical Stapler Cartridges to Inhaler Valves: An Example of Risk-Averse Production
An extensive, anonymized example highlights the effectiveness of a holistic quality system approach. For instance, there may be a program aimed at manufacturing valve components for a metered-dose inhaler (MDI). In such a component, accurate dosing is paramount for delivering medicine correctly. The first contractor delivered valve stems with satisfactory dimensional accuracy but variable sealing forces across lots, resulting in unreliable medicine dispensing.
1. The Issue: Insignificant Variability That Compromises Functionality
While the valve stems met the requirements for dimensionality specified in the drawings, their functionality — the opening and closing resistance force — proved inconsistent. It was found that this variability arose from micro-level differences in surface finish and edge geometry of a specific sealing land, factors unmeasured in conventional dimensional checks. Consequently, the product delivered either an inadequate or excessive amount of medicine to the patient.
2. The Systemic Solution: From Inspection to Process Control
An intervention came from the engineering team through a holistic approach. They started by redesigning the sealing land geometry with their new partner, ensuring manufacturability and consistency of this crucial feature. Second, they adopted 100% functional testing of the sealing force on a custom fixture, rather than sampling. Lastly, they developed SPC charts based on the critical machined dimensions (i.e., tool wear, feed rate), impacting the seal geometry. Thus, instead of finding defective parts and removing them, the company managed to prevent the creation of such defects.
3. The Quantifiable Outcome: Predictable Safety and Quality
Humanized Output: The result was really a groundbreaking one. The Cpk or process capability index soared over 1. 67 which indicated the sealing process was now very much capable and reliable. Device consistency in terms of performance escalated profoundly so that each inhaler would undoubtedly be delivering the correct dose. What is more, only a low and predictable scrap rate was achieved since the process control was achieved. It is clear that simply ensuring that a medical device meets certain requirements in order to reduce risks is not sufficient.
What is a True “Reliable Health Partnership” in Precision Manufacturing?
What is a true reliable health partnership? The answer is that it goes beyond the scope of a simple buyer and supplier transaction in manufacturing processes. Instead, it entails a strategic partnership based on mutual understanding and respect for the principles of excellence in patient safety and compliance with regulatory guidelines. Such a partner acts as a part of your quality management system, familiar with all the intricacies of work within the health sector. That is because there is a two-way exchange of quality data, integrated change management processes, and continuous deliveries of highly reliable and traceable parts. Hence, in the health technology industry, choosing a partner boils down to selecting an ally that shares your values.
1. Common Values and Regulatory Expertise: The basis of the relationship is the mutual understanding of what is at stake. The provider does not think about the order but the part that may affect the life of a patient. This principle governs all activities of the company. They are well versed in FDA 21 CFR Part 820, ISO 13485, and MDR/IVDR regulations and set up their processes accordingly, even exceeding the demands made by the regulators. Their approach is preventative, rather than reactive, as they constantly ask themselves how something could harm patients. Such a common vision makes your partner more than a supplier – they become your protector.
2. Integrated Quality Systems and Transparent Communication: In an effective partnership, the quality systems are highly integrated. The QMS from your partner must align with yours, making it easier to exchange documents, do audits, and solve problems together. Communication is not only open but also proactive and backed by solid data. They need to let you know of any problems that could arise beforehand and give you data to back up your own submission. The efficiency created by such integration can’t be understated.
3. Quality and Traceability throughout the Transition Process: Ultimately, our partnership ensures consistency and traceability in all processes. Whatever number of prototypes you need from us – whether it is five pieces for the purposes of verification testing or fifty thousand parts for the commercial release – we apply the same stringent principles of quality management, materials control, and inspections. Each part you receive will come with a comprehensive data package that explains everything about the item in question. The result is an entirely risk-free transition process with the assurance that the highest quality standards have been applied.
Conclusion
In the area of medical and health technologies, quality is directly tied to safety and effectiveness. In order to create a manufacturing supply chain that is not only efficient but also resilient, innovation teams should incorporate quality considerations into the initial design process, be demanding of stringent material traceability and environmental requirements, and carry out thorough audits of suppliers’ quality practices. All of this goes beyond mere compliance with regulations and represents the ultimate commitment to the safety and well-being of the users of health technology products.
H2: FAQs
Q: When ordering CNC turned components for a medical device, which tolerances should be given first consideration?
A: While basic sizes are considered, geometric tolerances such as cylindricity, concentricity, and true position are generally the most significant in achieving proper sealing and alignment. Since surface finish (Ra) is equally important, you must ensure that the tolerances are decided upon according to the performance criteria and not merely the tightening of standards on an arbitrary basis.
Q: How can I be sure that the CNC turned parts I am getting are completely biocompatible?
A: Biocompatibility is a system-wide feature so you need to get material certified with the relevant ISO 10993 test data for your type of device. Also, you should have full material test certificates including batch information able to be traced throughout the whole process from the initial raw material to the finished product. Moreover, the manufacturer needs to employ validated contamination-free procedures.
Q: What is the main difference between normal CNC turning and medical CNC turning?
A: The main difference is in the Quality Management System (QMS) where medical CNC turning operates a QMS that is certified to ISO 13485 and has design controls, validations, and change management requirements. Complete traceability and detailed documentation must be available for each step to ensure patient safety and quality standards are met.
Q: How fast can medical-grade CNC turned prototypes be delivered?
A: Lead times may range from 2 to 4 weeks, which is longer compared to the delivery time of regular products. The additional time is needed for careful process planning, certification of the material used, validation of inspection processes, and a complete first-article inspection. The detailed documentation is just as important as the actual parts in the process.
Q: What must I look for in an inspection report from the supplier regarding a turned medical part?
A: A full inspection report needs to contain: a complete FAIR with real measurements, material certification of the batch, certificates of conformance for any special processing, and any evidence for cleanliness validation. A good inspection report will be easily traced back, created according to the supplier’s QMS, and provide solid data for your design history file.
Author Bio
This author is an expert in manufacturing precise components for industries with the highest reliability demands and boasts 12 years of success working with life sciences and health technology organizations in creating supply chains which conform to the highest regulatory standards. This skill is utilized by the company LS Manufacturing, which offers complete manufacturing services ranging from design through to production of end-to-end traceable processes. The professionals work under a certified management system based on such standards as ISO 13485, IATF 16949, and AS9100D. For a thorough analysis and DFM compliance assessment of your device components, learn more about their extensive CNC turning services.



